In the Netherlands, the European Space Agency has built an experimental “oxygen plant” that can extract oxygen trapped within simulated Moon dust.
The process not only extracts up to 96 percent of the oxygen in the imitation lunar soil, but it also leaves behind metals that could be useful in future crewed missions to the Moon, Mars, and beyond.
Oxygen on the Moon
Scientists know that the Moon’s soil, or regolith, contains 40 to 45 percent oxygen by weight based on lunar rock samples returned to Earth during the Apollo missions. This makes oxygen the most abundant element on the Moon’s surface. However, harvesting it is difficult.

Beth Lomax/University of Glasgow
“Being able to acquire oxygen from resources found on the moon would obviously be hugely useful for future lunar settlers, both for breathing and in the local production of rocket fuel,” said Beth Lomax, a University of Glasgow researcher involved in the project, in an ESA press release.
We all understand the importance of breathable oxygen, but liquid oxygen is also used as a propellant in many rockets. Future missions would not have to fill up their rockets with a massive surplus of heavy liquid propellants if they could take oxygen directly from the Moon, a process known as in-situ resource utilization. This would essentially allow us to use the Moon as a cosmic gas station, significantly lowering the cost of far-reaching space missions to Mars and beyond.
Liquid oxygen could also be extremely useful for keeping satellites in orbit around the Earth. According to a paper published in the journal Planetary and Space Science by Lomax and colleagues, transporting liquid oxygen from the Moon to Earth-orbiting satellites is more energy efficient than launching oxygen from Earth’s surface while fighting our planet’s strong gravity.
The problem with extracting oxygen from lunar rocks is that it is bound up in chemical compounds, which require energy to separate. This calls for special methods of separating oxygen from the other elements in lunar regolith.
Splitting Rocks
To separate the oxygen from other components in faux Moon rocks, the researchers use a technique called molten salt electrolysis.
The researchers can let chemistry and physics do the heavy lifting by first putting the powdery Moon dust into a hot batch of molten calcium chloride salt, then running an electrical current through the mixture. The oxygen previously trapped in the simulated rocks migrates to an electrode (specifically, an anode), where it can be captured by the researchers.
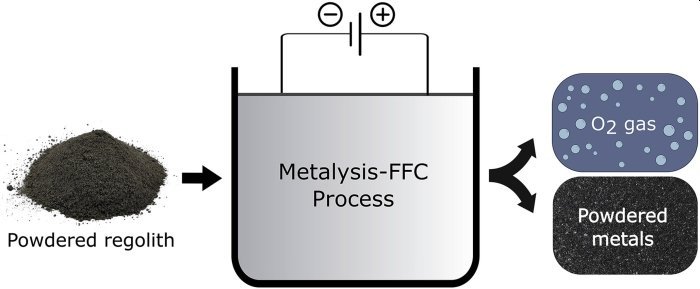
The researchers claim that they were able to extract 96 percent of the oxygen from their imitation Moon rocks in just 50 hours using this technique. Alternatively, in the first 15 hours, they can extract 75% of the oxygen. Furthermore, as an added bonus, the process produces a mixture of metal alloys that researchers believe could be useful as well.
In case you were wondering, researchers at NASA’s Kennedy Space Center are also working on a technique for harvesting oxygen from Moon rocks, according to a press release issued last August. So stay tuned, because in the not-too-distant future, we might have an old-fashioned gas station bidding war break out on the Moon.
READ MORE: The Moon’s Top Layer Alone Has Enough Oxygen To Sustain 8 Billion People For 100,000 Years






